Biochemical Data Constraining the S4 of Hv
In this post I will finish my series on alignments and homology models. Here, I will discuss three different biochemical studies of the voltage-gated proton channel (Hv) that help to delineate the boundaries of the S4 helix. First, I will discuss the structure of the coiled-coil, which limits where along the primary the sequence of the channel the S4 helix must end (Li et al., 2010). Next, I will discuss S4 truncation experiments that identify the functional limits of the S4 helix (Sakata et al., 2010). Finally, I will discuss accessibility studies that have been done on the S4 helix (Sakata et al., 2010; González et al. 2010). Although these data are not conclusive as to the exact boundaries of the S4 helix, taken together they can be used to rule out certain alignments between voltage-sensor domains (VSDs) that have nevertheless been used to construct homology models.
—
The Structure of the Coiled-coil
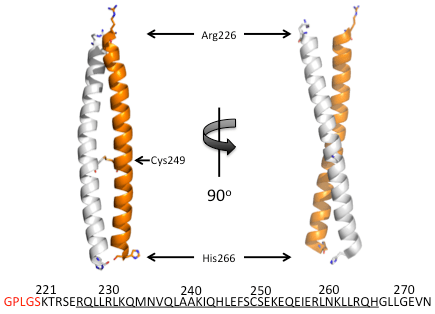
Figure 1. Cartoon representation of the structure of the coiled-coil from human Hv1. The coils, one from each of the two subunits in the dimer, are colored white and orange respectively. The protein sequence of the construct used in crystallization is shown along the bottom. The residues shown in red are from the cloning and expression process and are not part of the human Hv1 sequence. The structured amino acid residues residues are underlined. The coiled-coil structure extends from Arg226 to His266 (side chains shown in stick representation). The disulfide bond formed by Cys249 is shown. All other side chains are not shown.
The structure of the coiled-coil from human Hv1 has been experimentally determined by X-ray crystallography (Li et al., 2010). The coiled-coil was expressed as a glutathione S-transferase (GST) fusion protein in Escherichia coli (Li et al. 2009) and was purified with a glutathione affinity resin, after which the GST was removed by protease treatment (Li et al. 2009). This resulted in a coiled-coil protein with five additional non-native amino acid residues on the N-terminus (colored red in the sequence in Fig.1). Final enrichment was accomplished by ion exchange and size exclusion chromatography (Li et al. 2009).
The structure of the coiled-coil of human Hv1 is not very surprising or interesting: it looks like a coiled-coil (Fig.1). However, we can infer a few interesting observations from the structure . Firstly, the disulfide bond formed by cysteine 249, which was known to cross-link full-length human Hv1 dimers in cell membranes (Lee et al., 2008), is present and clearly visible in the structure (Fig.1). Next, the construct used in the crystallization has native amino acid residues that are not structured, on both the N– and C-termini (underlined vs. not-underlined residues of the sequence in Fig.1). This indicates that, although these residues are present in the protein, they are not part of the coiled-coil structure. This allows us to clearly demarcate the structured coiled-coil residues along the sequence of human Hv1 (underlined residues in Fig.1; see my previous post “Human Hv1: Architectural Overview” for the entire human Hv1 sequence). For example, the structured region of the coiled-coil ends at histidine 266 even though seven more amino acid residues are present in the protein (residues glycine 267 to asparagine 273, Fig.1). This indicates that these seven residues are unstructured in the native protein, flopping around in the cytosol. More interestingly, we can get an idea for where the coiled-coil begins. The first structured amino acid residues of the coiled-coil is arginine 226; however, the crystallized construct contains five native human Hv1 amino acid residues preceding arginine 226 (lysine 221 to glutamate 225). If these preceding residues were part of the coiled-coil in the full length channel, it is likely that we would see them adopting the coiled-coil structure here. Knowing exactly at which amino acid residue the coiled-coil structure begins in the human Hv1 sequence can help us define a limit on the structured region of the final transmembrane helix S4.

Figure 2. A selection of possible alignments between the S4 helices of Kv1.2-2.1 paddle chimera and human Hv1. The gating charge residues are shown in red. The amino acid residues of the S4 helix from the structure of Kv1.2-2.1 paddle chimera are boxed in orange. The exact boundaries of the human Hv1 S4 helix are unknown. However, the beginning of the coiled-coil structure is known and is underlined. The alignments are named according to the convention in Wood et al., 2011. Although only sequential alignments are shown above remember that it is possible that gap alignments may provide a better model for aligning S4 helices. Please see my post ”Is the S4 Helix of Hv Short?” for further discussion on this topic.
Fig.2 shows four of many possible alignments between the S4 helices of Kv1.2-2.1 paddle chimera and human Hv1. Although only sequential alignments are shown above, remember that it is possible that gap alignments may provide a better model for aligning S4 helices. As I have discussed previously in my posts “The Problem of Aligning S4” and “Is the S4 Helix of Hv Short?“, any data that help us constrain the limits of the S4 helix of Hv channels can help us narrow down the possible alignments between VSDs and give us a better understanding of the differences in the sequences and mechanisms of the different channels.
For instance, the structure of the coiled-coil shows us that arginine 226 is the first residue in the coiled-coil and therefore definitely not part of S4. Hence, any alignment that overlaps the S4 of the Kv1.2-2.1 paddle chimera with arginine 226 can be ruled out. To the best of my knowledge, no such alignment has been proposed. However, one of the most common alignments used to build homology models— alignment R1-Hv1 in Fig.2— comes pretty close. In this alignment, arginine 223 (only three residues up from the beginning of the coiled-coil structure) is located within S4 (please see models built in Ramsey et al,. 2010 and Wood et al., 2011).
It seems to me that the very short two-aminoacid-residue linker between S4 and the coiled-coil suggested by the models built using this alignment would not allow sufficient flexibility for the channel to undergo the conformational changes necessary for gating. Admittedly, nobody knows exactly what those conformational changes look like. However, it is clear from accessibility studies that significant conformational changes occur (see below), and it is unlikely that a two-aminoacid-residue linker would be able to accommodate them.
Going forward, it is clear that we can use the structure of the coiled-coil as a “yardstick” to estimate where the S4 helix of Hv is likely to end and to rule out any S4 alignments that clearly overlap with this structure.
—
Truncation of the S4 helix
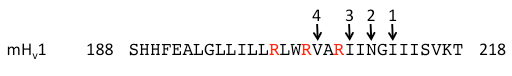
Figure 3. Functional truncations of mouse Hv1. The sequence of the wild type mouse Hv1 S4 helix is shown. The final amino acid residues of the truncated constructs are indicated by an arrow and are numbered from longest to shortest. The S4 arginines are shown in red.
A series of C-terminally truncated constructs of mouse Hv1 channels in which the coiled-coil and part of the putative S4 helix were removed have been generated and tested for function (Fig.3; Sakata et al., 2010). The most fascinating aspect of the four truncation constructs shown in Fig.3 is that they all generated functional voltage-gated proton channels! This is especially surprising for truncation construct 4, in which a significant portion of the putative S4 helix (including the third S4 arginine) is removed (Fig.3).
These data can be interpreted in two ways: if the most popular alignments used for building homology models (alignments R1-Hv1 and R2-Hv1 in Fig.2) are correct, then removing the C-terminal half of the S4 helix of Hv channels (i.e. leaving only the top half of the transmembrane helix) results in functional channels. On the other hand, if the most popular alignments are wrong, then perhaps these truncations don’t remove significant portions of the S4 helix. Given the importance of the S4 helix to the function of voltage sensors, it seems more probable to me that alignments R1-Hv1 and R2-Hv1, for which I have yet to find a convincing justification, are incorrect. This would lead us to consider more seriously the R3-Hv1 and R4-Hv1 alignments (Fig.2), for which truncation construct 4 would only remove a turn or two from the bottom of the S4 helix.
—
Accessibility Studies of S4

Figure 4. S4 helix accessibility studies. Sequences of the human (hHv1), mouse (mHv1) and ciona intestinalis (ciHv1) S4 helices are compared to the sequence of the Shaker Kv1 channel S4 helix. In the hHv1 sequence, histidine 193 is highlighted in blue because it is known to be exposed to the extracellular medium in the closed conformation of the channel. In the mHv1 sequence the residues highlighted in blue are accessible whereas the residues highlighted in green are buried in the closed conformation of the channel. In the ciHv1 and Shaker K sequences the amino acid residues are highlighted according to the key shown at the bottom of the figure. The residues highlighted in orange in the ciHv1 sequence are buried in both the open and closed states of the channel. The S4 arginines are shown in red for each sequence and numbered for the Shaker Kv1 sequence.
Fig.4 summarizes a lot of data generated using site-directed mutagenesis and cysteine-reactive chemical reagents to test the accessibility of specific positions along the S4 helix while the channel is in the open or closed state.
It is possible to determine whether a residue at a given position in the transmembrane helix is exposed to the aqueous media or buried in the membrane by mutating the amino acid to cysteine, adding a reagent that reacts with exposed cysteines and examining whether the two in fact react (indicating that the cysteine is exposed). These sorts of accessibility studies have been previously carried out on the Shaker Kv1 channel S4 helix (Larsson et al., 1996). What was determined for this channel was that the exposure of different positions along the S4 helix depended on whether the VSD was in the depolarized (open) or hyperpolarized (closed) conformation. (Although the conformations of the VSD are not necessarily coupled to the conformation of the pore— i.e. depolarized inactivated states exist in which the VSD is in the depolarized conformation but the channel is closed— I will refer to the depolarized and hyperpolarized conformations of the VSD as open and closed respectively for ease of comparison with the Hv VSD conformations) . Some amino acid residues were always exposed to the extracellular medium regardless of what conformation the channel was in (called “out” and highlighted in bright green on the Shaker sequence in Fig.4). These positions are clearly at the top (extracellular) side of the S4 helix. Similarly, other amino acid residues were always exposed to the intracellular medium (called “in” and highlighted in pink on the Shaker sequence Fig.4). These positions are clearly at the bottom (intracellular) side of the S4 helix. More interestingly, some positions were exposed only when the VSD was in the depolarized (open) state and buried when the VSD was in the hyperpolarized (closed) state (called “open-state dependent” and highlighted in dark green on the Shaker sequence Fig.4). In addition, some positions were exposed only when the VSD was in the hyperpolarized (closed) state and buried when the VSD was in the depolarized (open) state (called “close-state dependent” and highlighted in purple on the Shaker sequence Fig.4). Together, these data indicate that the VSDs undergo a significant conformational change upon gating that drastically alters the accessibility of amino acid residues along the S4 helix. Furthermore, these data can be used to help define the limits of the S4 helix: if you compare Fig.4 with Fig.2, you can see that the “out” and the “in” positions agree fairly well with the known structural limits of the S4 helix.
Similar studies have been carried out on Hv channels. However, it is already possible to identify one extracellularly exposed position on the human Hv1 S4 in the closed state without any mutagenesis. This residue is histidine 193 (highlighted in blue in Fig.4), which is known to be involved in the binding of extracellular Zn2+ when the channel is in the closed conformation (Ramsey et al., 2006). Therefore, histidine 193 must be exposed to the extracellular medium in the closed state. It is highly likely that this residue remains exposed when the channel is in the open state, making it similar to the bright green “out” positions of Shaker. However, to my knowledge, the accessibility of this position in the open conformation has not been experimentally verified.
Next, the experiments of Sakata et al. probed the accessibility of amino acid residue positions along the entire length of the S4 helix of mouse Hv1 in the closed conformation (Fig.4; Sakata et al., 2010). This extensive mutagenesis study completely defines the boundaries between the exposed (highlighted in light blue on the mHv1 sequence in Fig.4) and buried (highlighted in light green on the mHv1 sequence in Fig.4) positions. Human Hv1 likely shows a very similar accessibility pattern due to its homology with mouse Hv1: the two channels have nearly identical S4 helix amino acid sequences (Fig.4).
Finally, we need to consider the work of González et al. in which the accessibility of positions along the Ciona intestinalis Hv1 S4 helix is probed in both the open and closed conformations (González et al. 2010). González et al. were able to identify three positions that were accessible in the open state but buried in the closed state (called “open-state dependent” and highlighted in dark green on the ciHv1 sequence in Fig.4), two positions that were buried in both the open and closed states of the channel (highlighted in orange on the ciHv1 sequence in Fig.4) and two positions that were buried in the open state but accessible in the closed state (called “close-state dependent” and highlighted in purple on the ciHv1 sequence Fig.4). These data agree perfectly with those of Sakata et al. on the accessible positions along the S4 helix of the mouse Hv1 in the closed conformation (compare the highlighting on the mHv1 sequence with that of the ciHv1 sequence in Fig.4).
All together, these data give us significant insight into the S4 helix of Hv channels. The data clearly indicate that the conformational change between the closed and open states of Hv channels is very different from the equivalent VSD conformational change in Shaker. This difference is most evident when looking at the accessibility of the “middle” positions along the S4 helix. In Shaker, there is a very quick transition from the open-state dependent accessibility of arginine 365 (R2) and the closed-state dependent accessibility of arginine 368 (R3) and none of the amino acid positions tested were buried in both the open and closed states of the VSD. This suggests that the conformational movement of S4 likely generates a deep aqueous cleft within the VSD.
On the other hand, Hv channels show that roughly two full turns of the S4 helix remain buried in both the open and closed conformations. This difference clearly indicates that water accessibility is much more restricted along the S4 helix of Hv than along that of the Shaker channel. However, it is also clear that a significant conformational change does occur between Hv open and closed states, changing the accessibility of several amino acid residue positions on both the extracellular and intracellular sides of the S4 helix. As noted above, it is likely that this significant conformational change requires a longer linker between the bottom of the S4 helix and the coiled-coil structure than is provided with the most commonly used R1-Hv and R2-Hv alignments for construction of homology models (Fig.2).
The differences between the accessibilities of amino acid positions along the S4 helices of Shaker and Hv channels further reinforce the problems with trying to align these helices. The positions of the S4 helix sequences in Fig.4 represent a rough alignment based on the accessibility data. Of course, it is clear that many other different alignments are possible, especially when also considering the different possible gap alignments. However, looking at these data we can certainly rule out some of the many possible alignments— I have difficulty finding any biochemical support for the most commonly used R1-Hv and R2-Hv alignments (Fig.2). In fact, I think the data suggest that the R3-Hv and R4-Hv alignments are closer to the true alignment. No studies using homology models based on the the R3-Hv and R4-Hv alignments have been published so far.
—
Conclusion
By examining the biochemical data that exist on the S4 helix of Hv channels, we are able to constrain the S4 helix and eliminate certain potential alignments between the S4 helices of different VSDs. The structure of the coiled-coil of human Hv1 constrains the bottom of the S4 helix: residues that are in the coiled-coil certainly will not be in S4 and, due to the conformational changes that occur during channel gating, there is likely a several-residue “buffer” between the bottom of S4 and the start of the coiled-coil. The functional truncations of mouse Hv1 tell us that either Hv channels can function with only half of their S4 transmembrane helix or that the S4 helix doesn’t extend much beyond the final S4 arginine residue. Finally, studies of the accessibility of amino acid positions along the S4 helix tell us how the conformational change of Hv channels differs from that of the Shaker Kv channel. Together, all these data provide significant evidence that the most commonly used R1-Hv and R2-Hv alignments for the construction of homology models of human Hv1 are wrong. Therefore, we have to be extremely skeptical about any conclusions on Hv channel mechanism that are generated using these homology models.
Can we use the above data to predict the extent of the S4 helix of human Hv1? I think so, and Fig.5 represents my best guess, based on the biochemical data.

Figure 5. My best guess as to the extent of the S4 helix for human Hv1. The residues contained within the orange box constitute S4. The S4 arginines are shown in red. The beginning of the coiled-coil structure is underlined.
At the top (N-terminal end) of S4, my prediction contains all of the residues that are buried in the closed conformation plus a couple of additional “wiggle-room residues” that may or may not be part of the helix. Histidine 193 is exposed in the closed conformation and I place it in the S3-S4 linker. For the bottom of S4, my prediction encompasses all of the residues that become buried in the open conformation plus the additional isoleucines (I). It is possible that S4 ends at the flexible glycine (G) residue, but I thought ending it on the serine (S) was appropriate, since the side chain of serine residues are known to cap the ends of helices. Of course, without a structure of human Hv1 we can’t be sure which amino acid residues constitute the exact boundaries of the S4 helix.
What do you think? Is my prediction of the S4 at all accurate? Can you think of a better S4 helix for human Hv1? Do you agree that the biochemical evidence doesn’t support the use of the R1-Hv and R2-Hv alignments for the construction of homology models? If you agree or disagree I would love to know what you think. Also if you would like me to elaborate on any particular point I would be happy to do so. Please leave your questions and comments below.
—
Works Cited and Further Reading
Alabi, A. A., Bahamonde, M. I., Jung, H. J., Kim, J. I., & Swartz, K. J. (2007). Portability of paddle motif function and pharmacology in voltage sensors. Nature, 450(7168), 370–375. doi:10.1038/nature06266
González, C., Koch, H. P., & Larsson, H. P. (2010). Strong cooperativity between subunits in voltage-gated proton channels. Nature structural & molecular biology, 17(1), 51–56. doi:10.1038/nsmb.1739
Larsson, H. P., Baker, O. S., Dhillon, D. S., & Isacoff, E. Y. (1996). Transmembrane movement of the shaker K+ channel S4. Neuron, 16(2), 387–397.
Lee, S.-Y., Letts, J. A., & Mackinnon, R. (2008). Dimeric subunit stoichiometry of the human voltage-dependent proton channel Hv1. Proceedings of the National Academy of Sciences of the United States of America, 105(22), 7692–7695. doi:10.1073/pnas.0803277105
Li, S. J., Zhao, Q., Zhou, Q., & Zhai, Y. (2009). Expression, purification, crystallization and preliminary crystallographic study of the carboxyl-terminal domain of the human voltage-gated proton channel Hv1. Acta Cryst (2009). F65, 279-281 [doi:10.1107/S1744309109003777], 1–3. doi:10.1107/S1744309109003777
Li, S. J., Zhao, Q., Zhou, Q., Unno, H., Zhai, Y., & Sun, F. (2010). The Role and Structure of the Carboxyl-terminal Domain of the Human Voltage-gated Proton Channel Hv1. Journal of Biological Chemistry, 285(16), 12047–12054. doi:10.1074/jbc.M109.040360
Ramsey, I. S., Mokrab, Y., Carvacho, I., Sands, Z. A., Sansom, M. S. P., & Clapham, D. E. (2010). An aqueous H+ permeation pathway in the voltage-gated proton channel Hv1. Nature structural & molecular biology, 1–9. doi:10.1038/nsmb.1826
Ramsey, I. S., Moran, M. M., Chong, J. A., & Clapham, D. E. (2006). A voltage-gated proton-selective channel lacking the pore domain. Nature, 440(7088), 1213–1216. doi:10.1038/nature04700
Sakata, S., Kurokawa, T., Norholm, M. H. H., Takagi, M., Okochi, Y., Heijne, Von, G., & Okamura, Y. (2010). Functionality of the voltage-gated proton channel truncated in S4. Proceedings of the National Academy of Sciences of the United States of America, 107(5), 2313–2318. doi:10.1073/pnas.0911868107
Xu, Y., Ramu, Y., & Lu, Z. (2010). A Shaker K+ Channel with a Miniature Engineered Voltage Sensor. Cell, 1–10. doi:10.1016/j.cell.2010.07.013
Comments
One Response to “Biochemical Data Constraining the S4 of Hv”Trackbacks
Check out what others are saying...[…] avid readers of this blog may recall, I weighed in on this topic last summer in a series of posts (The Problem of aligning S4, Is the S4 helix of Hv short? and Biochemical Data Constraining the S4 of Hv). In my final entry […]